Treatment(BNCT)
- HOME
- Cancer(Treatment)
- Treatment(BNCT)
- A New Era in the World’s First Clinical Application of BNCT: Southern Tohoku BNCT Research Center Leads the Way in Precision Cancer Treatment
A New Era in the World’s First Clinical Application of BNCT: Southern Tohoku BNCT Research Center Leads the Way in Precision Cancer Treatment
26.04.15
On March 19, 2026, the first clinical BNCT (Boron Neutron Capture Therapy) treatment in China was successfully carried out at the Boao Lecheng International Medical Tourism Pilot Zone in Hainan Province. The patient, who had advanced recurrent tongue cancer and came from Henan Province, recovered smoothly after a single irradiation session lasting only about 30 minutes and has already returned to daily life. This achievement marks not only an important starting point for BNCT clinical application in Hainan, but also a significant step in bringing cell-level precision cancer treatment onto the international stage.
What is BNCT?
BNCT is a highly selective form of radiation therapy. It works by first delivering a boron-containing drug that preferentially accumulates in cancer cells. When the tumor area is then exposed to neutrons, boron-10 undergoes a nuclear reaction that releases short-range particles, precisely destroying cancer cells while minimizing damage to surrounding normal tissues. For this reason, BNCT is widely regarded as one of the most promising forms of cell-level precision cancer therapy.
Three Key Advantages of BNCT
1. Cell-level precision with minimal impact on normal tissue
BNCT selectively targets cancer cells while sparing surrounding healthy tissue as much as possible.
2. Treatment is often completed in a single session
For many patients, just one irradiation session of approximately 30 to 60 minutes is required.
3. A new option for recurrent tumors
Because of its high precision, BNCT may offer a valuable treatment option for patients with recurrent tumors, and in some cases, re-irradiation may be possible even for those who have previously undergone radiation therapy.
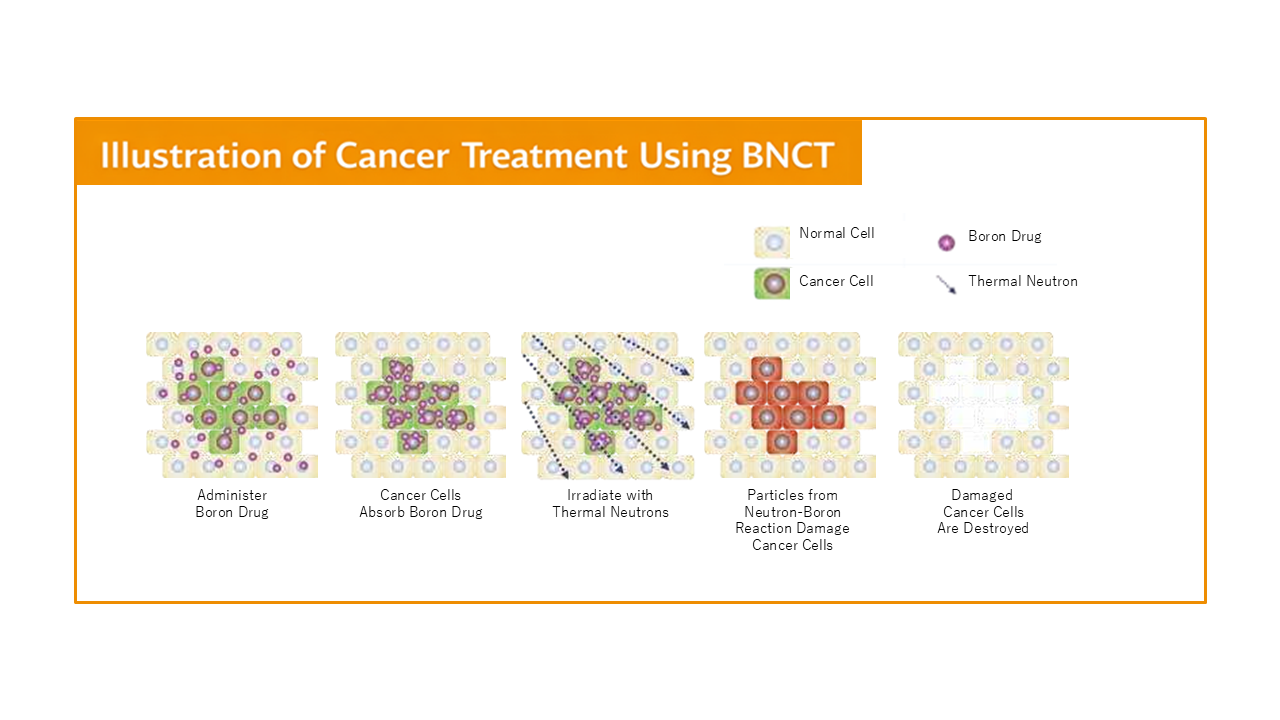
Japan has already entered the era of clinical use and insurance coverage
Japan has already approved BNCT for unresectable locally advanced or recurrent head and neck cancer, and the treatment is covered by the national health insurance system. In 2020, the accelerator-based BNCT system and the boron drug Steboronine® were approved. In May 2020, the Southern Tohoku BNCT Research Center became the first institution to provide BNCT as routine clinical treatment, and in June, the therapy became eligible for public insurance coverage, marking the beginning of a new era of standardized clinical use.

Southern Tohoku BNCT Research Center : At the forefront of the world
The Southern Tohoku BNCT Research Center is one of the world’s leading institutions advancing the clinical application of BNCT and has accumulated extensive clinical experience. As of February 2026, the center had treated more than 463 patients, placing it among Japan’s leading BNCT institutions.
Clinical data have also been encouraging. In a 2024 study of 47 patients, the complete response rate was 51%, the objective response rate was 74%, the 1-year overall survival rate was 86.1%, and the 2-year overall survival rate was 66.5%. In a 2026 meta-analysis that included 7 studies and 361 patients, the objective response rate for locally recurrent head and neck cancer was 70%, with a 2-year overall survival rate of 45%.
Rigorous evaluation and standardized treatment
BNCT is not suitable for every patient. Before treatment, FBPA-PET/CT is used to assess whether the boron-containing drug accumulates sufficiently in the tumor, and only eligible patients proceed to treatment. The center uses the NeuCure® system, developed by Sumitomo Heavy Industries and Kyoto University, and was the first institution in the world to use this device in formal clinical practice. BNCT treatment is usually completed in a single session, with a hospital stay of approximately 3 to 7 days, followed by 2 years of post-treatment follow-up.
The significance of Hainan’s first successful case
The completion of the first BNCT clinical treatment in Hainan marks the official launch of BNCT clinical practice in China and also reflects the deepening of international medical collaboration. With its advanced technology and extensive experience, the Southern Tohoku BNCT Research Center has become an important reference point for the development of BNCT worldwide. Since December 2024, the center has also begun accepting international patients, further promoting the global reach of BNCT.
BNCT is not only an advanced treatment technology, but also a source of new hope for patients with recurrent or difficult-to-treat cancers. The success of Hainan’s first case represents an important milestone in China’s BNCT clinical journey, while Japan’s Southern Tohoku BNCT Research Center continues to drive the field forward through its world-leading clinical experience.
※ This article is based on publicly available information from the Southern Tohoku BNCT Research Center and published medical literature.
